NEJM:发现治疗克罗恩氏病的潜在新药物疗法
2013-05-06 ZinFingerNase 生物谷
2012年10月19日优特克诺(ustekinumab, 也译作优斯它单抗)是一种能够治疗被称作银屑病的皮肤病的抗体。如今,根据一项最新的研究,来自美国加州大学圣地亚哥分校的研究人员发现它也能降低克罗恩氏病(Crohn's Disease)的破坏性影响。相关研究结果于2012年10月18日刊登在New England Journal of Medicine期刊上。 来自临床试验的结果证实优特克诺

2012年10月19日优特克诺(ustekinumab, 也译作优斯它单抗)是一种能够治疗被称作银屑病的皮肤病的抗体。如今,根据一项最新的研究,来自美国加州大学圣地亚哥分校的研究人员发现它也能降低克罗恩氏病(Crohn's Disease)的破坏性影响。相关研究结果于2012年10月18日刊登在New England Journal of Medicine期刊上。
来自临床试验的结果证实优特克诺增加中重度克罗恩氏病患者的临床反应和病情缓解。克罗恩氏病是一种炎症性肠病(inflammatory bowel disease, IBD),能够导致多种不好的症状,包括腹泻、肠道出血和体重减轻。这种疾病也能够导致严重性的并发症,如肠道梗阻和脓肿。
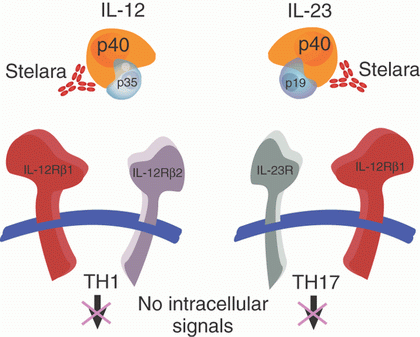
论文第一作者、加州大学圣地亚哥分校医学院肠胃病科主任William J. Sandborn博士说,“在治疗克罗恩氏病患者中,我们所面临的最大挑战是成功地治疗身体对肿瘤坏死因子(tumor necrosis factor, TNF)抑制剂如英利昔单抗(Remicade)、阿达木单抗(Humira)和赛妥珠单抗(Cimzia)产生抗性的患者。优特克诺阻断两种导致炎症的蛋白:白细胞介素-12 (IL-12)和白细胞介素-23 (IL-23)。这一发现是为这些患者开发出新的治疗方法而取得的一次重要进步。”
三分之一的中重度克罗恩氏病患者并不对当前利用TNF抑制剂进行的治疗方法产生反应,其中这些抑制剂调节着人体的免疫系统和炎症。还有三分之一的患者只产生临时的反应。
在12个国家开展的随机化临床试验中,研究人员以其中的526名患者作为研究对象。入选的患者年龄至少18岁,而且被诊断患有克罗恩氏病的时间不低于三个月。
在以安慰剂作为对照的研究中,患者接受为期36周的治疗。在研究开始时,这些患者被给予一次静脉注射优特克诺剂量,随后每八周,他们就被给予一次皮下注射剂量。早在接受这种疗法的6周时,研究人员就能够观察到这种治疗方法的益处。
在接受治疗的患者当中,研究人员报道,有5名患者患有严重性的感染,以及1名患者患有基底细胞癌。
如今,研究人员正在更多的临床试验来跟踪这些大有希望的初始研究结果,以便能够开发出一种疗法来增加这些患者对优特克诺的反应,缓解他们的病情和改善他们的生活质量。
与克罗恩氏病相关的拓展阅读:

Ustekinumab Induction and Maintenance Therapy in Refractory Crohn's Disease
BACKGROUND
In patients with Crohn's disease, the efficacy of ustekinumab, a human monoclonal antibody against interleukin-12 and interleukin-23, is unknown.
METHODS
We evaluated ustekinumab in adults with moderate-to-severe Crohn's disease that was resistant to anti–tumor necrosis factor (TNF) treatment. During induction, 526 patients were randomly assigned to receive intravenous ustekinumab (at a dose of 1, 3, or 6 mg per kilogram of body weight) or placebo at week 0. During the maintenance phase, 145 patients who had a response to ustekinumab at 6 weeks underwent a second randomization to receive subcutaneous injections of ustekinumab (90 mg) or placebo at weeks 8 and 16. The primary end point was a clinical response at 6 weeks.
RESULTS
The proportions of patients who reached the primary end point were 36.6%, 34.1%, and 39.7% for 1, 3, and 6 mg of ustekinumab per kilogram, respectively, as compared with 23.5% for placebo (P=0.005 for the comparison with the 6-mg group). The rate of clinical remission with the 6-mg dose did not differ significantly from the rate with placebo at 6 weeks. Maintenance therapy with ustekinumab, as compared with placebo, resulted in significantly increased rates of clinical remission (41.7% vs. 27.4%, P=0.03) and response (69.4% vs. 42.5%, P<0.001) at 22 weeks. Serious infections occurred in 7 patients (6 receiving ustekinumab) during induction and 11 patients (4 receiving ustekinumab) during maintenance. Basal-cell carcinoma developed in 1 patient receiving ustekinumab.
CONCLUSIONS
Patients with moderate-to-severe Crohn's disease that was resistant to TNF antagonists had an increased rate of response to induction with ustekinumab, as compared with placebo. Patients with an initial response to ustekinumab had significantly increased rates of response and remission with ustekinumab as maintenance therapy. (Funded by Janssen Research and Development; CERTIFI ClinicalTrials.gov number, NCT00771667.)
本网站所有内容来源注明为“梅斯医学”或“MedSci原创”的文字、图片和音视频资料,版权均属于梅斯医学所有。非经授权,任何媒体、网站或个人不得转载,授权转载时须注明来源为“梅斯医学”。其它来源的文章系转载文章,或“梅斯号”自媒体发布的文章,仅系出于传递更多信息之目的,本站仅负责审核内容合规,其内容不代表本站立场,本站不负责内容的准确性和版权。如果存在侵权、或不希望被转载的媒体或个人可与我们联系,我们将立即进行删除处理。
在此留言












#克罗恩氏病#
51
#药物疗法#
54
#新药物#
56